 (ii) relationship between number of hydrogen atoms and number of water molecules:įrom the molecular formula we see that 1 molecule of water is made up of 2 atoms of hydrogen and 1 atom of oxygen. N(H 2O molecules) = number of water molecules N(H 2O molecules) = n(H 2O molecules) × N A (i) relationship between moles of water molecules and number of water molecules is: Step 4: What is the relationship between moles of water molecules and number of hydrogen atoms? 
Step 3: What is the relationship between moles of particles and number of particles? N(H 2O molecules) = moles of water molecules = 1.5 mol N(H atoms) = number of hydrogen atoms = ? Determine the number of hydrogen atoms in 1.5 moles of water, H 2O, molecules. Number of ammonia molecules = 2.1 × 10 24 molecules (Note: 2 significant figures are justified) Step 5: Substitute in the vales and solve for N: Step 4: Write the equation for the relationship between between moles of ammonia molecules and number of ammonia molecules: Step 3: What is the relationship between the moles of particles and the number of particles? N = amount of ammonia molecules in moles = 3.5 mol Step 2: What information (data) has been given in the question? N(ammonia) = number of ammonia molecules = ? Step 1: What is the question asking you to do?Ĭalculate the number of ammonia molecules. Question 1: Calculate the number of ammonia, NH 3, molecules in 3.5 moles of ammonia. Step 5: Substitute in the vales and solve for the unknownĬalculating the number of particles (N = n × N A).Step 4: Write the equation for the specific relationship you need to solve the problem.Step 3: What is the general relationship between the moles of particles and the number of particles?.Step 2: What information (data) has been given in the question?.Step 1: What is the question asking you to do?.The solutions to these problems are given as a set of 6 general steps to help structure your approach to problem solving: Worked Examples: Moles-Avogadro Number Calculations Moles of atoms of element Y = n(Y) = (N × b) ÷ N AĬonsider the following examples in which 1.927 × 10 24 molecules of a compound with the general formula X 2Y are present Moles of atoms of element X = n(X) = (N × a) ÷ N A Number of atoms of element Y = N(Y) = N × b Number of atoms of element X = N(X) = N × a The equation n = N ÷ N A can also be used to find the amount in moles of atoms or ions in a compound if you know both the molecular formula for the compound and the number of molecules of the compound that are present.įor N molecules of a compound with the general formula X aY b: If you know a substance contains 3.011 × 10 23 particles of the substance, then the moles of substance will be (3.011 × 10 23) ÷ (6.022 × 10 23) = 0.5 molģ.011 × 10 23 helium atoms = 0.5 mol of helium atomsģ.011 × 10 23 sodium ions = 0.5 mol of sodium ionsģ.011 × 10 23 water molecules = 0.5 mol of water molecules 
The table below gives the moles of each element present in the compound, and also shows us how to calculate the number of atoms of each element present: (n × b) × 6.022 × 10 23 atoms of element YĬonsider n moles of each of these compounds with the general formula XY 2. 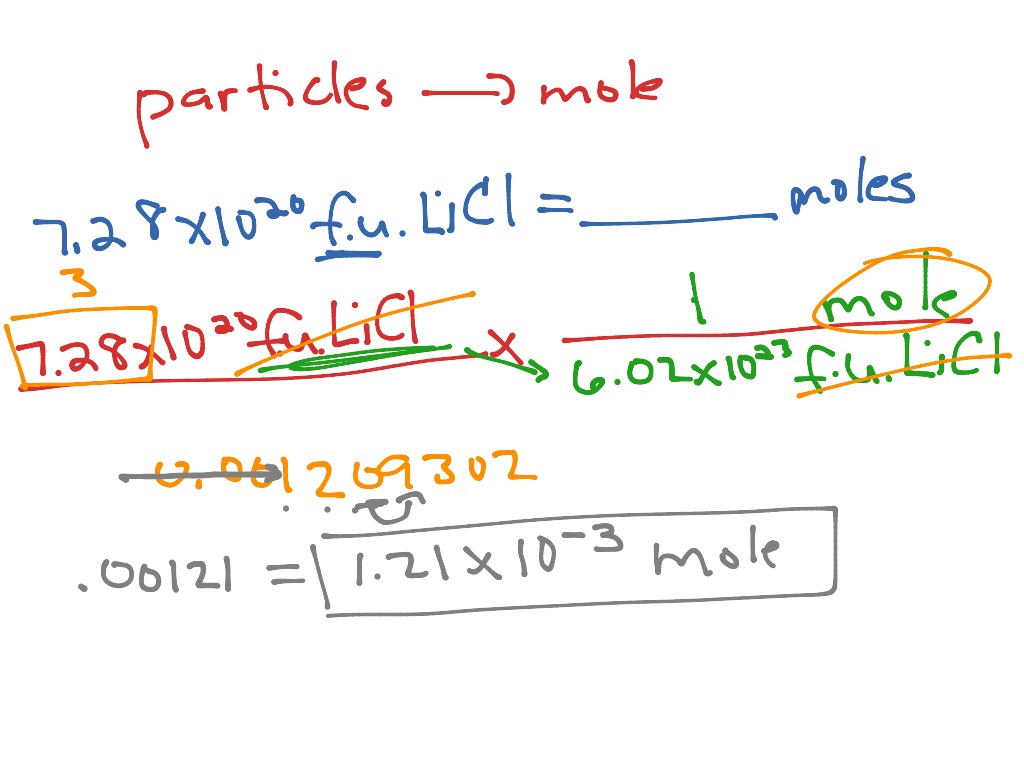
The mathematical equation, N = n × N A, can also be used to find the number of atoms of each element in a known amount (in moles) of a compound.įor a compound with the molecular formula X aY b: The mathematical equation, N = n × N A, can be used to find the number of atoms, ions or molecules in any amount (in moles) of atoms, ions or molecules: 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
